-
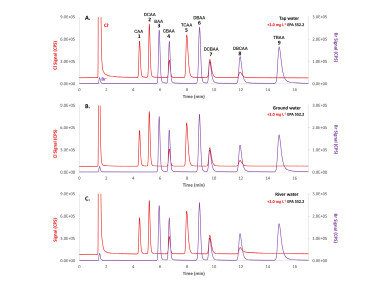 Figure 2: separation of haloacetic acids in tap (A), ground (B), and river water (C) samples spiked with the EPA 552.2 certified reference material [2].
Figure 2: separation of haloacetic acids in tap (A), ground (B), and river water (C) samples spiked with the EPA 552.2 certified reference material [2]. -
 Figure 1: Analysis of environmental contaminants in different water matrices by HPLC-ICPMS/MS
Figure 1: Analysis of environmental contaminants in different water matrices by HPLC-ICPMS/MS
HPLC, UHPLC
Analysis of environmental contaminants in different water matrices by HPLC-ICPMS/MS
Sep 24 2024
Halogenated acetic acids (HAAs) are among the most common water disinfection byproducts. Since these are presumably harmful to health, the US Environmental Protection Agency (EPA) regulates the levels of five haloacetic acids (monochloro-, dichloro-, trichloro-, monobromo- and dibromoacetic acid) to an overall maximum of 60 μg/L. The regulations of the European Union include another four haloacetic acids, with a maximum concentration of 80 μg/L for all nine HAAs combined [1].
High performance liquid chromatography coupled with electrospray ionisation tandem mass spectrometry (HPLC-ESI-MS/MS) is commonly used to achieve high sensitivity and selectivity. However, this detection technique is prone to matrix effects due to ion suppression or enhancement in the ionisation source. This application note will demonstrate the analysis of nine HAAs by using inductively coupled plasma tandem mass spectrometry (ICPMS/MS) for detection, which is less prone to matrix effects [2]. Different types of water samples from Austria were analysed using a YMC-Triart C18 column: tap water from Graz, groundwater from the Leutschach well and river water from the Mur.
Figure 1 shows the baseline separation of nine HAAs, each spiked with 2.0 mg/L EPA 552.2 reference material, in about 15 min. A high injection volume is used to achieve the lowest possible limit of detection (LOD). Therefore, the alkaline hard water samples (pH = 7.1–7.9) must be acidified to reduce the pH difference with the mobile phase.
The LOD ranges from 1.8–2.0 μg Cl/L and 1.0–1.5 μg Br/L, while the limit of quantification varies between 4.6 and 12 μg/L. The recoveries obtained show that ICPMS/MS detection is less prone to matrix effects compared to ESI-MS/MS because interfering ions with high carbon load (e.g. bicarbonate) are separated from the analytes as they elute with the dead volume. Therefore, an internal standard (IS) is not required unlike ESI-MS/MS, where an isotopically labelled IS for each analyte is preferable. Furthermore, ICPMS/MS detection can also be performed using non-volatile buffers containing phosphate, sulphate, chloride or sodium.
Find more informative applications on YMC’s homepage or download the YMC-Triart columns brochure for more details.
[1] European Parliament.Quality of water intended for human consumption ***I (accessed 08.2024)
[2] Bassam Lajin and Walter Goessler, Analytical Chemistry 2020 92 (13), 9156-9163, DOI: 10.1021/acs.analchem.0c01456.
Events
May 11 2025 Vienna, Austria
May 18 2025 Tempe. AZ, USA
May 21 2025 Birmingham, UK
Jun 01 2025 Baltimore, MD, USA
Jun 15 2025 Bruges, Belgium















